|
Calera’s process for CO2 reduction involves the capture of CO2 gas from industrial emitting sources and converting the CO2 into a novel calcium carbonate cement system that can be used to make a variety of valuable products. Calera’s focus is on the manufacture of building materials and more specifically, novel board products from Calera’s novel cement system. Calera’s process removes CO2 from emitting sources without having to concentrate the CO2 and converts the gas into a solid form of calcium carbonate thereby permanently sequestering the CO2. The Calera process is unique in that environmental sustainability is coupled with economic sustainability and is a CO2 capture and conversion technology that turns CO2 into a profitable feedstock.
CO2 + Alkalinity + Calcium = Calcium Carbonates
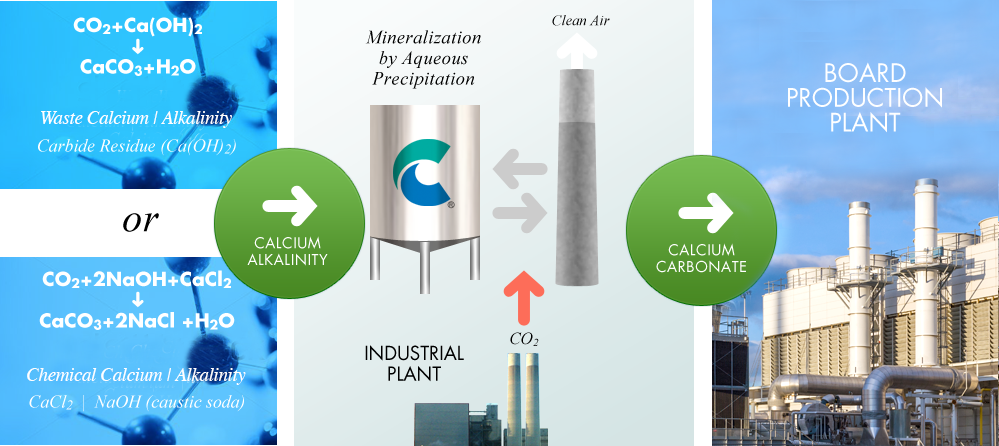
The conversion of CO2 to calcium carbonate requires a source of alkalinity and calcium. One option is the use of industrial waste streams that contain both alkalinity and calcium, for example in the form of calcium hydroxide (Ca(OH)2). Another option is to use alkalinity, such as caustic soda (NaOH), which can be supplied from the conventional industrial manufacturing process. Calera is also developing a proprietary, low energy technology for the production of caustic soda. Calcium, in the form of for example calcium chloride can be naturally occurring or can also be found in the waste streams of existing chemical processes. The Calera process can use either waste or chemical of waste inputs, both resulting in the capture and conversion of CO2 to the special calcium carbonate cement which can then be used to form board products. |

