|
Calera’s ABLE platform allows for the production of a wide range of commodity chemicals. To date, particular focus has been given to the production of caustic soda (NaOH), hydrogen (H2) and ethylene dichloride (C2H4Cl2).
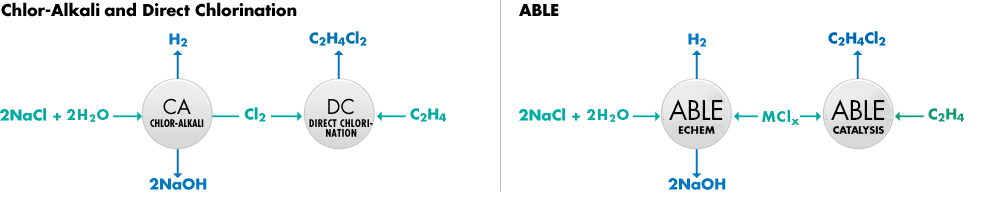
In the conventional production method, salt (NaCl) and water are electrochemically split through the Chlor-Alkali process to produce chlorine gas (Cl2), NaOH and H2. For this process, a significant portion of the required electrical energy is associated with the production of Cl2. This Cl2 then reacts with ethylene (C2H4) in the Direct Chlorination process to produce C2H4Cl2.
Calera’s ABLE process significantly reduces the overall energy associated with making NaOH, H2 and C2H4Cl2 compared with the conventional process. This is done by replacing the high energy Cl2 gas evolution with an innovative metal chloride shuttle reaction. In this way, Calera uses the same feedstocks to produce the same products at lower energy. This lower energy results in lower CO2 emissions as well as significant cost savings.
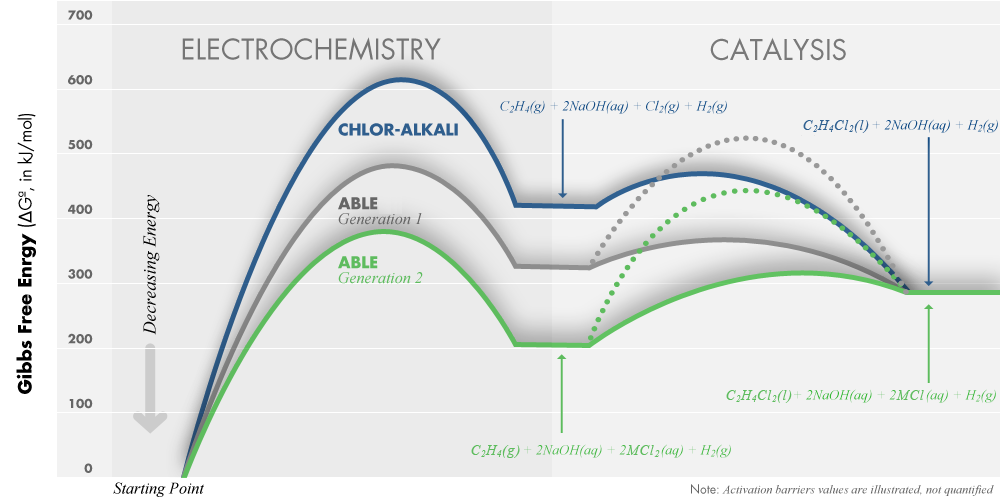
These energy savings can be better understood through a Free Energy diagram. Starting with the same feedstocks and producing the same final products, Calera’s process requires significantly lower energy to arrive at the intermediate reactants. Calera is currently in the process of scaling the ABLE technology to the pilot scale at its facilities in Moss Landing, California. |

